 Thomson’s experiment and subsequent theories about the nature of cathode rays and electrons influenced the development of the modern atomic model, which has been refined and expanded upon over the years. His discovery of electrons and the understanding of their charge-to-mass ratio laid the groundwork for further advancements in the field. Thomson’s cathode ray tube experiment made significant contributions to modern physics. Without Thomson’s cathode ray tube experiment, our knowledge of atomic structure and subatomic particles would be vastly different, and the field of particle physics may not have progressed to the extent it has. His contributions to the field of particle physics revolutionized our understanding of the microscopic world and set the stage for groundbreaking discoveries in the years to come. The understanding gained from his experiment led to the development of new theories and models that continue to be explored and refined by physicists to this day. Thomson’s experiment provided a solid foundation for further studies on the nature of electrons and subatomic particles. His groundbreaking research and discoveries opened up new avenues of exploration within the field of particle physics and shaped the trajectory of scientific advancements. 
Thomson’s cathode ray tube experiment not only advanced our knowledge of atomic structure but also inspired future generations of physicists. Emily Johnson, Physics Professor The Influence on Future Physicists His discovery of the charge-to-mass ratio of electrons laid the foundation for future advancements in the field of particle physics.” – Dr. “Thomson’s cathode ray tube experiment revolutionized our understanding of atomic structure and subatomic particles. 
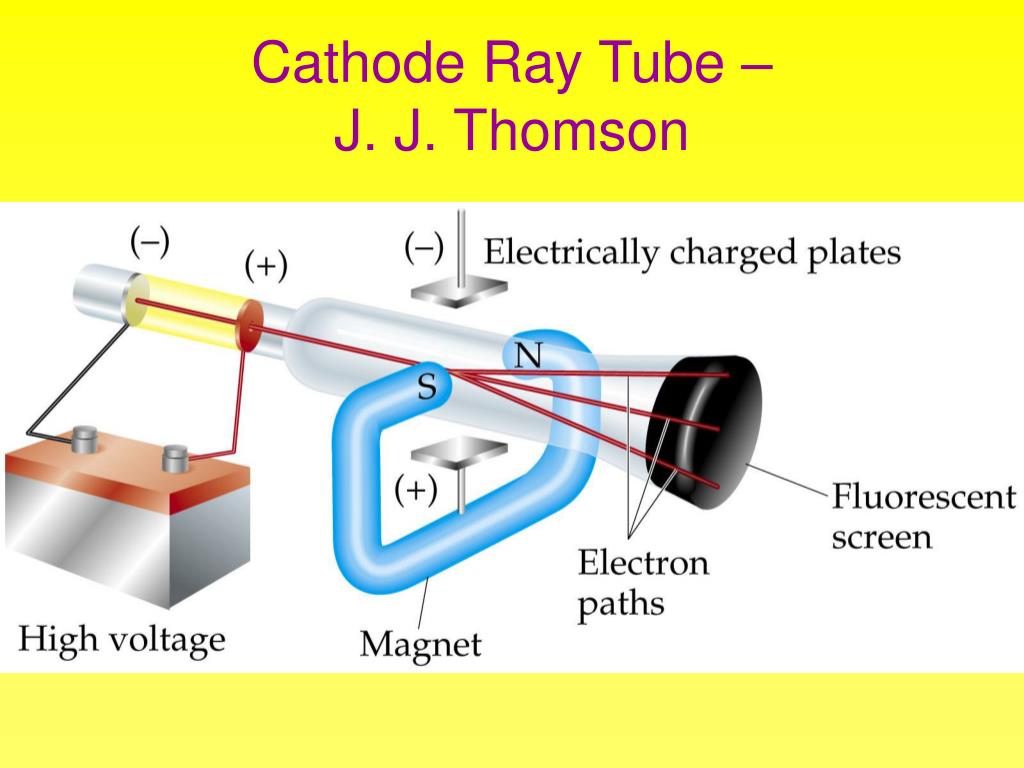
Thomson’s discovery of electrons and his contributions to modern physics solidify his place as one of the pioneers in the field. Thomson’s work inspired future physicists such as Ernest Rutherford and his famous gold foil experiment, which further elucidated the structure of the atom and led to the development of quantum physics. Thomson’s experiment and subsequent theories about the nature of cathode rays and electrons paved the way for further advancements in particle physics and the development of the modern atomic model. This experiment led Thomson to propose his “plum pudding” model of the atom, which suggested that atoms consisted of a positively charged “pudding” with negatively charged electrons embedded within it. 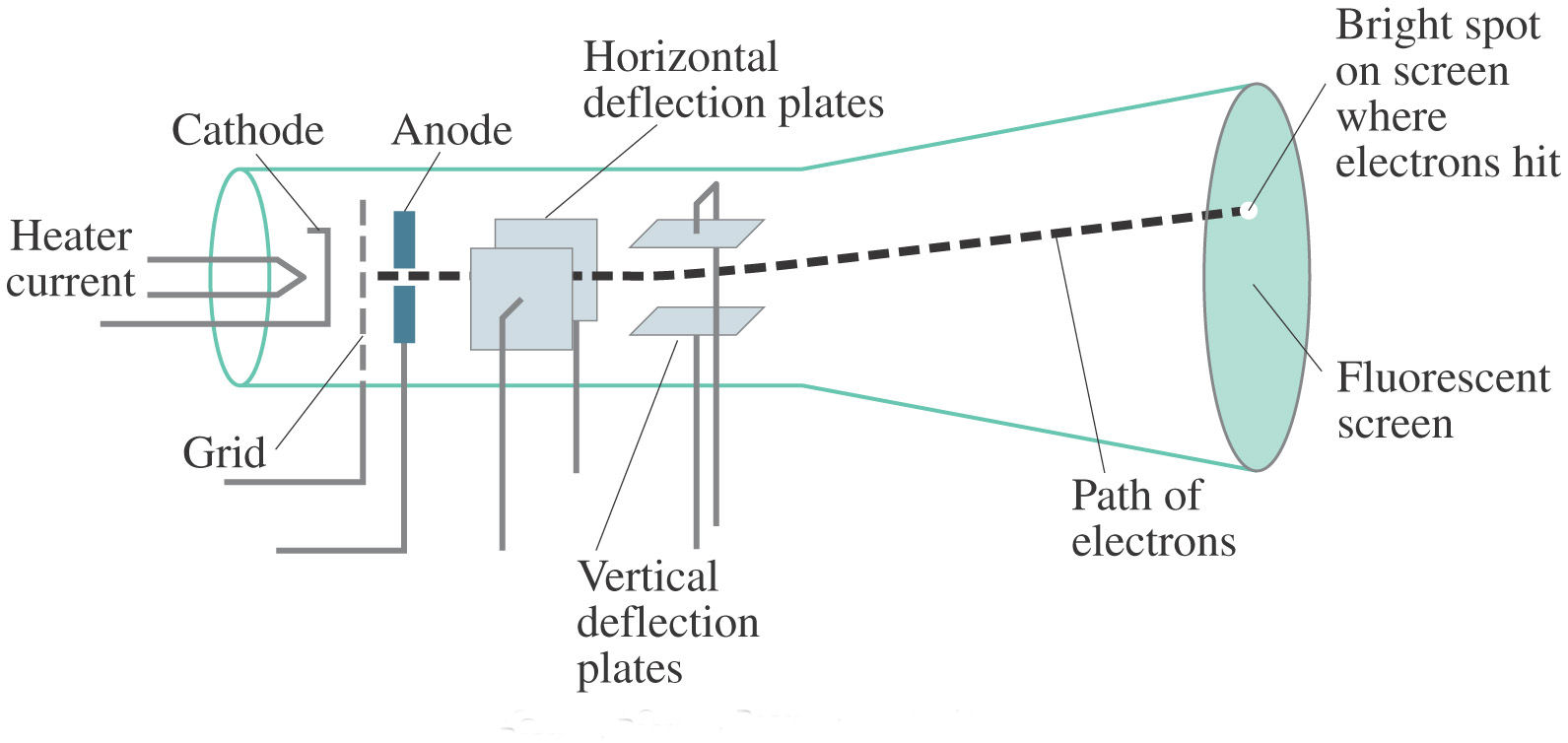
Thomson’s manipulation of cathode rays and observations of their movement and behavior allowed him to determine the charge-to-mass ratio of electrons. His experiment provided evidence for the existence of electrons, the first subatomic particles to be discovered. Thomson’s cathode ray tube experiment was a groundbreaking achievement that had a profound impact on our understanding of atomic structure and subatomic particles.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
